top of page
A Level Syllabus Content
(Adapted from Singapore Examinations and Assessment Board)
Introduction
Candidates will be assumed to have knowledge and understanding of Chemistry at O-Level, as a single subject or as part of a balanced science course. This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need for students to develop skills that will be of long term value in an increasingly technological world rather than focusing on large quantities of factual material which may have only short term relevance. Experimental work is an important component and should underpin the teaching and learning of Chemistry.
Aims
The aims of a course based on this syllabus should be to:
1. provide students with an experience that develops interest in Chemistry and builds the knowledge, skills and attitudes necessary for further studies in related fields
2. enable students to become scientifically literate citizens who are well-prepared for the challenges of the 21st century
3. develop in students the understanding, skills, ethics and attitudes relevant to the Practices of Science, including the following:
3.1 understanding the nature of scientific knowledge
3.2 demonstrating science inquiry skills
3.3 relating science and society
4. develop the way of thinking to explain phenomena, approach and solve problems in chemical systems which involves students in:
4.1 understanding the structure, properties and transformation of matter at the atomic/molecular level and how they are related to each other
4.2 connecting between the submicroscopic, macroscopic and symbolic levels of representations in explaining and making predictions about chemical systems, structures and properties.
Practices of Science
laws, and theories); it is a way of knowing and doing. It includes an understanding of the nature of scientific knowledge and how this knowledge is generated, established and communicated. Scientists rely on a set of established procedures and practices associated with scientific inquiry to gather evidence and test their ideas on how the natural world works. However, there is no single method and the real process of science is often complex and iterative, following many different paths. While science is powerful, generating knowledge
that forms the basis for many technological feats and innovations, it has limitations.
The Practices of Science are explicitly articulated in this syllabus to allow teachers to embed them as learning objectives in their lessons. Students’ understanding of the nature and limitations of science and scientific inquiry are developed effectively when the practices are taught in the context of relevant science content. Attitudes relevant to science such as inquisitiveness, concern for accuracy and precision, objectivity, integrity and perseverance should be emphasised in the teaching of these practices where appropriate. For
example, students learning science should be introduced to the use of technology as an aid in practical work or as a tool for the interpretation of experimental and theoretical results.
1. Understanding the Nature of Scientific Knowledge
1.1 Understand that science is an evidence-based, model-building enterprise concerned with the natural world
1.2 Understand that the use of both logic and creativity is required in the generation of scientific knowledge
1.3 Recognise that scientific knowledge is generated from consensus within the community of scientists through a process of critical debate and peer review
1.4 Understand that scientific knowledge is reliable and durable, yet subject to revision in the light of new evidence
2. Demonstrating Science Inquiry Skills
2.1 Identify scientific problems, observe phenomena and pose scientific questions/hypotheses
2.2 Plan and conduct investigations by selecting the appropriate experimental procedures, apparatus and materials, with due regard for accuracy, precision and safety
2.3 Obtain, organise and represent data in an appropriate manner
2.4 Analyse and interpret data
2.5 Construct explanations based on evidence and justify these explanations through reasoning and logical argument
2.6 Use appropriate models1 to explain concepts, solve problems and make predictions
2.7 Make decisions based on evaluation of evidence, processes, claims and conclusions
2.8 Communicate scientific findings and information using appropriate language and terminology
3. Relating Science and Society
3.1 Recognise that the application of scientific knowledge to problem solving could be influenced by other considerations such as economic, social, environmental and ethical factors
3.2 Demonstrate an understanding of the benefits and risks associated with the application of science to society
3.3 Use scientific principles and reasoning to understand, analyse and evaluate real-world systems as well as to generate solutions for problem solving
Curriculum Framework
The key features of the H2 Chemistry Curriculum comprise Core Ideas and Extension Topics, Practices of
Science and Learning Experiences as illustrated in Fig. 1.

1. Core Ideas and Extension Topics
The topics in H2 Chemistry are organised as two levels underpinned by the Practices of Science:
(a) Core ideas: The three Core Ideas of Chemistry are Matter, Structure and Properties, and Transformation. The concepts in these Core Ideas are inter-related and form the basis for which further learning and understanding of chemical phenomena and reactions is built upon.
(b) Extension topics: Concepts in the Core Ideas extend into the learning of different chemical systems such as chemistry of organic compounds and transition elements. As an example, an understanding of concepts of Chemical Bonding and The Periodic Table is extended to the study of the chemistry of transition metals where students learn to appreciate the similarities and differences when comparing with main group metals.
2. Practices of Science
The Practices of Science are common to the natural sciences of physics, chemistry and biology. These practices highlight the ways of thinking and doing that are inherent in the scientific approach, with the aim of equipping students with the understanding, skills, and attitudes shared by the scientific disciplines, including an appropriate approach to ethical issues.
3. Learning Experiences
The Learning Experiences2 refer to a range of learning opportunities selected by teachers to link the chemistry content with the Core Ideas and the Practices of Science to enhance students’ learning of the concepts. Rather than being mandatory, teachers are encouraged to incorporate Learning Experiences that match the interests and abilities of their students and provide opportunities to illustrate and exemplify the Practices of Science, where appropriate. Real-world contexts can help illustrate the concepts in chemistry
and their applications. Experimental activities and ICT tools can also be used to build students’ understanding.
Assessment Objectives
The Assessment Objectives listed below reflect those parts of the Aims and Practices of Science that will be assessed.
(A) Knowledge with understanding
Candidates should be able to demonstrate knowledge and understanding in relation to:
1. scientific phenomena, facts, laws, definitions, concepts and theories
2. scientific vocabulary, terminology and conventions (including symbols, quantities and units)
3. scientific instruments and apparatus, including techniques of operation and aspects of safety
4. scientific quantities and their determination
5. scientific and technological applications with their social, economic and environmental implications.
The syllabus content defines the factual knowledge that candidates may be required to recall and explain. Questions testing these objectives will often begin with one of the following words: define, state, name, describe, explain or outline (see the Glossary of Terms).
(B) Handling, applying and evaluating information
Candidates should be able (in words or by using symbolic, graphical and numerical forms of presentation) to:
1. locate, select, organise and present information from a variety of sources
2. handle information, distinguishing the relevant from the extraneous
3. manipulate numerical and other data and translate information from one form to another
4. analyse and evaluate information so as to identify patterns, report trends and conclusions, and draw inferences
5. present reasoned explanations for phenomena, patterns and relationships
6. apply knowledge, including principles, to novel situations
7. bring together knowledge, principles, concepts and skills from different areas of chemistry, and apply them in a particular context
8. evaluate information and hypotheses
9. construct arguments to support hypotheses or to justify a course of action
10. demonstrate an awareness of the limitations of Chemistry theories and models.
These Assessment Objectives cannot be precisely specified in the syllabus content because questions testing such skills may be based on information which is unfamiliar to the candidate. In answering such questions, candidates are required to use principles and concepts that are within the syllabus and apply them in a logical, reasoned or deductive manner to a novel situation. Questions testing these objectives will often begin with one of the following words: predict, suggest, construct, calculate or determine (see the Glossary of Terms).
(C) Experimental skills and investigations
Candidates should be able to:
1. follow a detailed set or sequence of instructions and use techniques, apparatus and materials safely and effectively
2. make, record and present observations and measurements with due regard for precision and accuracy
3. interpret and evaluate observations and experimental data
4. identify a problem, devise and plan investigations, select techniques, apparatus and materials
5. evaluate methods and techniques, and suggest possible improvements.
Scheme of Assessment
All candidates are required to enter for Papers 1, 2, 3 and 4.

Paper 1 (1 h, 30 marks)
This paper consists of 30 compulsory multiple choice questions. Five to eight items will be of the multiple completion type.
All questions will include 4 options.
Paper 2 (2 h, 75 marks)
This paper consists of a variable number of structured questions including data-based questions. All questions are compulsory and answered on the question paper. The data-based question(s) constitute(s) 20–25 marks for this paper. The data-based question(s) provide(s) good opportunity to test higher order thinking skills such as handling, applying, and evaluating information. Some questions will also require candidates to integrate knowledge and understanding from different areas and topics of the chemistry syllabus.
Paper 3 (2 h, 80 marks)
This paper consists of two sections:
● Section A worth 60 marks consisting 3–4 free response questions, all compulsory. Each question constitutes 15–25 marks.
● Section B worth 20 marks consisting two questions, each of 20 marks. Candidates are to answer any one question.
These questions will require candidates to integrate knowledge and understanding from different areas and topics of the chemistry syllabus.
Paper 4 (2 h 30 min, 55 marks)
This paper will assess appropriate aspects of objectives C1 to C5 in the following skill areas:
● Planning (P)
● Manipulation, measurement and observation (MMO)
● Presentation of data and observations (PDO)
● Analysis, conclusions and evaluation (ACE)
The assessment of Planning (P) will have a weighting of 5%. The assessment of skill areas MMO, PDO and ACE will have a weighting of 15%. The scope of the practical paper is indicated in the Practical Assessment section. The assessment of PDO and ACE may also include questions on data-analysis which do not require practical equipment and apparatus.
Candidates will not be permitted to refer to books and laboratory notebooks during the assessment.
Weighting of Assessment Objectives

Additional Information
Data Booklet
A Data Booklet is available for use in the theory papers. The booklet is reprinted at the end of this syllabus document. The Qualitative Analysis Notes in the Data Booklet will be made available for use in the practical assessment.
Nomenclature
Candidates will be expected to be familiar with the nomenclature used in the syllabus. The proposals in "Signs, Symbols and Systematics" (The Association for Science Education Companion to 16–19 Science, 2000) will generally be adopted although the traditional names sulfate, sulfite, nitrate, nitrite, sulfurous and nitrous acids will be used in question papers. Sulfur (and all compounds of sulfur) will be spelt with f (not with ph) in question papers, however candidates can use either spelling in their answers.
Units and significant figures Candidates should be aware that misuse of units and/or significant figures, i.e. failure to quote units where
necessary, the inclusion of units in quantities defined as ratios or quoting answers to an inappropriate number of significant figures, is liable to be penalised.
Disallowed Subject Combinations
Candidates may not simultaneously offer Chemistry at H1 and H2 levels.
Content Map

Chemistry is about the study of matter, its interactions and transformations. At a macroscopic level, we observe matter and its interactions everywhere in our daily life. The submicroscopic level looks at the structure of matter that gives rise to these interactions. At O-Level, students have been introduced to the fundamental idea that matter is made up of particles and the simple atomic model (electrons in discrete shells around a positively charged nucleus). This allows students to apply the key ideas of conservation of
matter and energy in the quantitative treatment of reactions such as stoichiometry and thermochemistry.
At A-Level, an in-depth study of the electronic structure of atoms provides the basis for the study of chemical bonding. The Valence Shell Electron Pair Repulsion (VSEPR) model is used to visualise the threedimensional structure of molecules, which determines the type of interactions possible and also helps to explain the physical and chemical properties. Knowledge of structure and bonding is also important to study and predict trends in properties of matter and its reactions. Modern organic chemistry is based on the
concept that the four valence bonds of a carbon atom are oriented in a tetrahedral configuration, which led to the development of the stereochemical concept. This has important applications, particularly in the pharmaceutical industry as many drugs are active only in one particular enantiomeric form.
Transformation of matter involves the study of the feasibility (thermodynamics and kinetics considerations) and extent of chemical reactions (concept of equilibrium). The chemical thermodynamics dimension builds upon prior knowledge of thermochemistry, mainly enthalpy changes (ΔH). Together with entropy changes (ΔS), a mathematical relationship is established which gives the true measure of the thermodynamics feasibility of a reaction – the Gibbs free energy (ΔG). For aqueous redox reactions, the more convenient
notion of electrode potential (E) is used, and the resultant cell potential (Ecell) gives a measure of thermodynamics feasibility instead. The chemical kinetics facet of a reaction can be understood quantitatively by relating the rate of reaction to concentration of reactants. The qualitative aspect which deals with the factors affecting rate of reactions will be covered based on the collision theory.
The concepts in chemical thermodynamics and kinetics will form the basis for the study of Chemical Equilibrium. Theoretically all reactions are reversible, and the notion of dynamic equilibrium will be introduced. The concept of equilibrium constant (K) is understood via the equilibrium expression, which gives a measure of the extent of a reversible reaction. Factors which determine the position of equilibrium will also be examined. Chemical equilibria in aqueous media involving acids and bases will be dealt with in greater
depth, in view of the relevance and prevalence of these concepts which cuts across chemical systems.
This curriculum framework provides students the opportunity to appreciate the connections between the concepts in the Core Ideas of Matter, Structure and Properties, and Transformation, and to apply these to the study of different chemical systems in the Extension Topics.
Subject Content
CORE IDEA 1 – MATTER
1. Atomic Structure
Content
● The nucleus of the atom: neutrons and protons, isotopes, proton and nucleon numbers
● Electrons: electronic energy levels, ionisation energies, atomic orbitals, extranuclear structure
Learning Outcomes
Candidates should be able to:
(a) identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses
(b) deduce the behaviour of beams of protons, neutrons and electrons in an electric field
(c) describe the distribution of mass and charges within an atom
(d) deduce the numbers of protons, neutrons and electrons present in both atoms and ions given proton and nucleon numbers (and charge)
(e) (i) describe the contribution of protons and neutrons to atomic nuclei in terms of proton number and nucleon number
(ii) distinguish between isotopes on the basis of different numbers of neutrons present
(f) describe the number and relative energies of the s, p and d orbitals for the principal quantum numbers 1, 2 and 3 and also the 4s and 4p orbitals
(g) describe the shapes of s, p and d orbitals (see also Section 13) [knowledge of wave functions is not required]
(h) state the electronic configuration of atoms and ions given the proton number (and charge)
(i) explain the factors influencing the ionisation energies of elements (see the Data Booklet) (see also Section 5)
(j) deduce the electronic configurations of elements from successive ionisation energy data
(k) interpret successive ionisation energy data of an element in terms of the position of that element within the Periodic Table
CORE IDEA 2 – STRUCTURE AND PROPERTIES
2. Chemical Bonding
Content
• Ionic bonding, metallic bonding, covalent bonding and co-ordinate (dative covalent) bonding
• Shapes of simple molecules and bond angles
• Bond polarities and polarity of molecules
• Intermolecular forces, including hydrogen bonding
• Bond energies and bond lengths
• Lattice structure of solids
• Bonding and physical properties
Learning Outcomes
Candidates should be able to:
(a) show understanding that all chemical bonds are electrostatic in nature and describe:
(i) ionic bond as the electrostatic attraction between oppositely charged ions
(ii) covalent bond as the electrostatic attraction between a shared pair of electrons and positively charged nuclei
(iii) metallic bond as the electrostatic attraction between a lattice of positive ions and delocalised electrons
(b) describe, including the use of ‘dot-and-cross’ diagrams,
(i) ionic bonding as in sodium chloride and magnesium oxide
(ii) covalent bonding as in hydrogen; oxygen; nitrogen; chlorine; hydrogen chloride; carbon dioxide; methane; ethene
(iii) co-ordinate (dative covalent) bonding, as in formation of the ammonium ion and in the Al2Cl6 molecule
(c) describe covalent bonding in terms of orbital overlap (limited to s and p orbitals only), giving σ and π bonds (see also Section 11.1)
(d) explain the shapes of, and bond angles in, molecules such as BF3 (trigonal planar); CO2 (linear); CH4 (tetrahedral); NH3 (trigonal pyramidal); H2O (bent); SF6 (octahedral) by using the Valence Shell Electron Pair Repulsion theory
(e) predict the shapes of, and bond angles in, molecules analogous to those specified in (d)
(f) explain and deduce bond polarity using the concept of electronegativity [quantitative treatment of electronegativity is not required]
(g) deduce the polarity of a molecule using bond polarity and its molecular shape (analogous to those specified in (d));
(h) describe the following forces of attraction (electrostatic in nature):
(i) intermolecular forces, based on permanent and induced dipoles, as in CHCl3(l); Br2(l) and the liquid noble gases
(ii) hydrogen bonding, using ammonia and water as examples of molecules containing –NH and –OH groups
(i) outline the importance of hydrogen bonding to the physical properties of substances, including ice and water
(j) explain the terms bond energy and bond length for covalent bonds
(k) compare the reactivities of covalent bonds in terms of bond energy, bond length and bond polarity
(l) describe, in simple terms, the lattice structure of a crystalline solid which is:
(i) ionic, as in sodium chloride and magnesium oxide
(ii) simple molecular, as in iodine
(iii) giant molecular, as in graphite and diamond
(iv) hydrogen-bonded, as in ice
(v) metallic, as in copper
[the concept of the ‘unit cell’ is not required]
(m) describe, interpret and/or predict the effect of different types of structure and bonding on the physical properties of substances
(n) suggest the type of structure and bonding present in a substance from given information
3. The Gaseous State
Content
• Ideal gas behaviour and deviations from it
• pV = nRT and its use in determining a value for Mr
• Dalton’s Law and its use in determining the partial pressures of gases in a mixture
Learning Outcomes
Candidates should be able to:
(a) state the basic assumptions of the kinetic theory as applied to an ideal gas
(b) explain qualitatively in terms of intermolecular forces and molecular size:
(i) the conditions necessary for a gas to approach ideal behaviour
(ii) the limitations of ideality at very high pressures and very low temperatures
(c) state and use the general gas equation pV = nRT in calculations, including the determination of Mr
(d) use Dalton’s Law to determine the partial pressures of gases in a mixture (see also Section 9)
4. Theories of Acids and Bases
Content
• Arrhenius, Brønsted-Lowry and Lewis theories of acids and bases
Learning Outcomes
Candidates should be able to:
(a) show understanding of, and apply the Arrhenius theory of acids and bases
(b) show understanding of, and apply the Brønsted-Lowry theory of acids and bases, including the concept of conjugate acids and conjugate bases
(c) show understanding of, and apply the Lewis theory of acids and bases (including non-aqueous system, e.g. reaction between BF3 and NH3)
5. The Periodic Table
Content
● Periodicity of atomic and physical properties of the elements: variation with proton number across the third period (sodium to chlorine) and down the group (Group 2 and Group 17) of:
(i) electronic configuration
(ii) atomic radius and ionic radius
(iii) ionisation energy
(iv) electronegativity
(v) melting point
(vi) electrical conductivity
• Periodicity of chemical properties of the elements in the third period:
(i) variation in oxidation number and bonding of the oxides (sodium to sulfur only) and of the chlorides (sodium to phosphorus only)
(ii) reactions of these oxides and chlorides with water
(iii) acid/base behaviour of these oxides and the corresponding hydroxides
• Periodicity of chemical properties of the elements down the group (Group 2 and Group 17):
(i) as reducing agents (Group 2) and oxidising agents (Group 17)
(ii) thermal stability of Group 2 carbonates and Group 17 hydrides
Learning Outcomes
Trends and variations in atomic and physical properties
For elements in the third period (sodium to chlorine), and in Group 2 (magnesium to barium) and Group 17 (chlorine to iodine) candidates should be able to:
(a) recognise variation in the electronic configurations across a Period and down a Group
(b) describe and explain qualitatively the general trends and variations in atomic radius, ionic radius, first ionisation energy and electronegativity:
(i) across a Period in terms of shielding and nuclear charge
(ii) down a Group in terms of increasing number of electronic shells, shielding and nuclear charge
(c) interpret the variation in melting point and in electrical conductivity across a Period in terms of structure and bonding in the elements (metallic, giant molecular, or simple molecular)
(d) describe and explain the trend in volatility of the Group 17 elements in terms of instantaneous dipoleinduced dipole attraction
Trends and variations in chemical properties
For elements in the third period (sodium to chlorine) candidates should be able to:
(e) (i) state and explain the variation in the highest oxidation number of the elements in oxides (for Na2O; MgO; Al2O3; SiO2; P4O10; SO3) and chlorides (for NaCl; MgCl2; AlCl3; SiCl4; PCl5)
(ii) state and explain the variation in bonding in oxides and chlorides in terms of electronegativity (with the exception of AlCl3)
(iii) describe the reactions of the oxides with water (for Na2O; MgO; Al2O3; SiO2; P4O10; SO3)
(iv) describe and explain the acid/base behaviour of oxides (for Na2O; MgO; Al2O3; SiO2; P4O10; SO3) and hydroxides (for NaOH; Mg(OH)2; Al(OH)3), including, where relevant, amphoteric behaviour in reaction with sodium hydroxide (only) and acids
(v) describe and explain the reactions of the chlorides with water (for NaCl; MgCl2; AlCl3; SiCl4; PCl5)
(vi) suggest the types of structure and bonding present in the oxides and chlorides from observations of their chemical and physical properties
For elements in Group 2 (magnesium to barium) and Group 17 (chlorine to iodine) candidates should be able to:
(f) describe and deduce from E⦵ values the relative reactivity of elements of:
(i) Group 2 as reducing agents
(ii) Group 17 as oxidising agents
(g) describe and explain the trend in thermal stability of:
(i) Group 2 carbonates in terms of the charge density of the cation and the polarisability of the large anion
(ii) Group 17 hydrides in terms of bond energies
In addition, candidates should be able to:
(h) predict the characteristic properties of an element in a given Group by using knowledge of chemical periodicity
(i) deduce the nature, possible position in the Periodic Table, and identity of unknown elements from given information of physical and chemical properties
CORE IDEA 3 – TRANSFORMATION
6. The Mole Concept and Stoichiometry
Content
• Relative masses of atoms and molecules
• The mole, the Avogadro constant
• The calculation of empirical and molecular formulae
• Reacting masses and volumes (of solutions and gases)
Learning Outcomes [the term relative formula mass or Mr will be used for ionic compounds]
Candidates should be able to:
(a) define the terms relative atomic, isotopic, molecular and formula mass
(b) define the term mole in terms of the Avogadro constant
(c) calculate the relative atomic mass of an element given the relative abundances of its isotopes
(d) define the terms empirical and molecular formula
(e) calculate empirical and molecular formulae using combustion data or composition by mass
(f) write and/or construct balanced equations
(g) perform calculations, including use of the mole concept, involving:
(i) reacting masses (from formulae and equations)
(ii) volumes of gases (e.g. in the burning of hydrocarbons)
(iii) volumes and concentrations of solutions
[when performing calculations, candidates’ answers should reflect the number of significant figures given or asked for in the question]
(h) deduce stoichiometric relationships from calculations such as those in (g)
7. Chemical Energetics: Thermochemistry and Thermodynamics (Gibbs Free Energy and Entropy)
Content
• Enthalpy changes: ΔH, of formation; combustion; hydration; solution; neutralisation; atomisation; bond energy; lattice energy; electron affinity
• Hess’ Law, including Born-Haber cycles
• Entropy and Free Energy
Learning Outcomes
Candidates should be able to:
(a) explain that most chemical reactions are accompanied by energy changes, principally in the form of heat usually associated with the breaking and forming of chemical bonds; the reaction can be exothermic (ΔH negative) or endothermic (ΔH positive)
(b) construct and interpret an energy profile diagram, in terms of the enthalpy change of the reaction and of the activation energy (see also Section 8)
(c) explain and use the terms:
(i) enthalpy change of reaction and standard conditions, with particular reference to: formation; combustion; hydration; solution; neutralisation; atomisation
(ii) bond energy (ΔH positive, i.e. bond breaking) (see also Section 2)
(iii) lattice energy (ΔH negative, i.e. gaseous ions to solid lattice)
(d) calculate enthalpy changes from appropriate experimental results, including the use of the relationship: heat change = mcΔT
(e) explain, in qualitative terms, the effect of ionic charge and of ionic radius on the numerical magnitude of a lattice energy
(f) apply Hess’ Law to construct simple energy cycles, e.g. Born-Haber cycle, and carry out calculations involving such cycles and relevant energy terms (including ionisation energy and electron affinity), with particular reference to:
(i) determining enthalpy changes that cannot be found by direct experiment, e.g. an enthalpy change of formation from enthalpy changes of combustion
(ii) the formation of a simple ionic solid and of its aqueous solution
(iii) average bond energies
(g) explain and use the term entropy
(h) discuss the effects on the entropy of a chemical system by the following:
(i) change in temperature
(ii) change in phase
(iii) change in the number of particles (especially for gaseous systems)
(iv) mixing of particles
[quantitative treatment is not required]
(i) predict whether the entropy change for a given process or reaction is positive or negative
(j) state and use the equation involving standard Gibbs free energy change of reaction, ΔG⦵: ΔG⦵ = ΔH⦵ – TΔS⦵
[the calculation of standard entropy change, ΔS⦵, for a reaction using standard entropies, S⦵, is not
required]
(k) state whether a reaction or process will be spontaneous by using the sign of ΔG⦵
(l) understand the limitations in the use of ΔG⦵ to predict the spontaneity of a reaction
(m) predict the effect of temperature change on the spontaneity of a reaction, given standard enthalpy and entropy changes
8. Reaction Kinetics
Content
● Simple rate equations; orders of reaction; rate constants
● Concept of activation energy
● Effect of concentration, temperature, and catalysts on reaction rate
● Homogeneous and heterogeneous catalysis
● Enzymes as biological catalysts
Learning Outcomes
Candidates should be able to:
(a) explain and use the terms: rate of reaction; rate equation; order of reaction; rate constant; half-life of a reaction; rate-determining step; activation energy; catalysis
(b) construct and use rate equations of the form rate = k[A]m[B]n (limited to simple cases of single-step reactions and of multi-step processes with a rate-determining step, for which m and n are 0, 1 or 2), including:
(i) deducing the order of a reaction by the initial rates method
(ii) justifying, for zero- and first-order reactions, the order of reaction from concentration-time graphs
(iii) verifying that a suggested reaction mechanism is consistent with the observed kinetics
(iv) predicting the order that would result from a given reaction mechanism
(v) calculating an initial rate using concentration data [integrated forms of rate equations are not required]
(c) (i) show understanding that the half-life of a first-order reaction is independent of concentration
(ii) use the half-life of a first-order reaction in calculations
(d) calculate a rate constant using the initial rates method
(e) devise a suitable experimental technique for studying the rate of a reaction, from given information
(f) explain qualitatively, in terms of frequency of collisions, the effect of concentration changes on the rate of a reaction
(g) show understanding, including reference to the Boltzmann distribution, of what is meant by the term activation energy
(h) explain qualitatively, in terms both of the Boltzmann distribution and of collision frequency, the effect of temperature change on a rate constant (and hence, on the rate) of a reaction
(i) (i) explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of lower activation energy, giving a larger rate constant
(ii) interpret this catalytic effect on a rate constant in terms of the Boltzmann distribution
(j) outline the different modes of action of homogeneous and heterogeneous catalysis, including:
(i) the Haber process
(ii) the catalytic removal of oxides of nitrogen in the exhaust gases from car engines (see also Section 11.3)
(iii) the catalytic role of atmospheric oxides of nitrogen in the oxidation of atmospheric sulfur dioxide
(iv) catalytic role of Fe2+ in the I–/S2O82– reaction
(k) describe enzymes as biological catalysts which may have specific activity
(l) explain the relationship between substrate concentration and the rate of an enzyme-catalysed reaction in biochemical systems
9. Chemical Equilibria
Content
● Chemical equilibria: reversible reactions; dynamic equilibrium
(i) factors affecting chemical equilibria
(ii) equilibrium constants
(iii) the Haber process
Learning Outcomes
Candidates should be able to:
(a) explain, in terms of rates of the forward and reverse reactions, what is meant by a reversible reaction and dynamic equilibrium
(b) state Le Chatelier’s Principle and apply it to deduce qualitatively (from appropriate information) the effects of changes in concentration, pressure or temperature, on a system at equilibrium
(c) deduce whether changes in concentration, pressure or temperature or the presence of a catalyst affect the value of the equilibrium constant for a reaction
(d) deduce expressions for equilibrium constants in terms of concentrations, Kc, and partial pressures, Kp [treatment of the relationship between Kp and Kc is not required]
(e) calculate the values of equilibrium constants in terms of concentrations or partial pressures from appropriate data
(f) calculate the quantities present at equilibrium, given appropriate data (such calculations will not require the solving of quadratic equations)
(g) show understanding that the position of equilibrium is dependent on the standard Gibbs free energy change of reaction, ΔG⦵
[Quantitative treatment is not required]
(h) describe and explain the conditions used in the Haber process, as an example of the importance of an understanding of chemical equilibrium in the chemical industry
EXTENSION TOPICS
10. Chemistry of Aqueous Solutions
10.1 Acid-base Equilibria
This topic deals with acid-base equilibria in aqueous solution. Hence the Brønsted-Lowry definitions of acid/base are primarily used in the understanding of pH of solutions. (See Section 4)
Content
● Acid dissociation constants, Ka and the use of pKa
● Base dissociation constants, Kb and the use of pKb
● The ionic product of water, Kw
● pH: choice of pH indicators
● Buffer solutions
Learning Outcomes
Candidates should be able to:
(a) explain qualitatively the differences in behaviour between strong and weak acids and bases in terms of the extent of dissociation
(b) explain the terms pH; Ka; pKa; Kb; pKb; Kw and apply them in calculations, including the relationship Kw = KaKb
(c) calculate [H+(aq)] and pH values for strong acids, weak monobasic (monoprotic) acids, strong bases, and weak monoacidic bases
[Calculations involving weak acids/bases will not require solving of quadratic equations]
(d) describe the changes in pH during acid-base titrations and explain these changes in terms of the strengths of the acids and bases
(e) explain the choice of suitable indicators for acid-base titrations, given appropriate data
(f) (i) explain how buffer solutions control pH
(ii) describe and explain their uses, including the role of H2CO3/HCO3– in controlling pH in blood
(g) calculate the pH of buffer solutions, given appropriate data
10.2 Solubility Equilibria
Content
• Solubility product; the common ion effect and complex ion formation
Learning Outcomes
Candidates should be able to:
(a) show understanding of, and apply, the concept of solubility product, Ksp
(b) calculate Ksp from concentrations and vice versa
(c) discuss the effects on the solubility of ionic salts by the following:
(i) common ion effect
(ii) formation of complex ion, as exemplified by the reactions of halide ions with aqueous silver ions followed by aqueous ammonia (see also Section 13)
11. Organic Chemistry
Preamble
Although there are features of organic chemistry topics that are distinctive, it is intended that appropriate cross-references with other sections/topics in the syllabus should be made. In their study of organic chemistry, candidates may wish to group the organic reactions in terms of the mechanisms in the syllabus where possible. Candidates may wish to compare and contrast the different mechanisms.
When describing preparative reactions, candidates will be expected to quote the reagents, e.g. aqueous NaOH, the essential practical conditions, e.g. reflux, high temperature and pressure, and the identity of each of the major products. Detailed conditions involving specific temperature and pressure values are not required.
Detailed knowledge of practical procedures is also not required; however, candidates may be expected to suggest (from their knowledge of the reagents, essential conditions and products) what steps may be needed to purify/extract a required product from the reaction mixture. In equations for organic redox reactions, the symbols [O] and [H] are acceptable.
11.1 Introduction
Content
• Empirical, molecular and structural formulae
• Functional groups and the naming of organic compounds
• Common terms for organic reactions and reactivities
• Shapes of organic molecules; σ and π bonds
Candidates are expected to be able to interpret and use the following types of representations in the description of organic molecules. The examples given are for the compound (+)-lactic acid. Empirical Formula: simplest ratio of the number of atoms of the elements present in one molecule, e.g. CH2O
Molecular Formula: actual number of atoms of the elements present in one molecule, e.g. C3H6O3 Structural Formula: shows how the constituent atoms of a molecule are joined together with minimal detail, using conventional groups, for an unambiguous structure, e.g. CH3CH(OH)CO2H Full Structural or Displayed Formula: detailed structure of molecule showing the relative placing of atoms
and the number of bonds between them, e.g.

Skeletal Formula: simplified representation of an organic formula derived from the structural formula by removing hydrogen atoms (and their associated bonds) and carbon atoms from alkyl chains, leaving just the carbon-carbon bonds in the carbon skeleton and the associated functional groups
Skeletal or partial skeletal representations may be used in question papers and are acceptable in
candidates’ answers where they are unambiguous, e.g.

Stereochemical Formula: show spatial arrangement of bonds, atoms and groups in molecule in 3-D, e.g.

When drawing a pair of optical isomers, candidates should indicate the three-dimensional structures according to the convention used in the example below.

Candidates are expected to be able to interpret and use the curly arrow notation to represent the movement of electrons in organic reaction mechanisms.

Learning Outcomes
Candidates should be able to:
(a) interpret, and use the nomenclature, general formulae and displayed formulae of the following classes of compound:
(i) hydrocarbons (alkanes, alkenes and arenes)
(ii) halogen derivatives (halogenoalkanes and halogenoarenes)
(iii) hydroxyl compounds (alcohols and phenols)
(iv) carbonyl compounds (aldehydes and ketones)
(v) carboxylic acids and derivatives (acyl chlorides and esters)
(vi) nitrogen compounds (amines, amides, amino acids and nitriles)
(b) interpret, and use the following terminology associated with organic reactions:
(i) functional group
(ii) degree of substitution: primary, secondary, tertiary, quaternary
(iii) homolytic and heterolytic fission
(iv) carbocation
(v) free radical, initiation, propagation, termination
(vi) electrophile (Lewis acid), nucleophile (Lewis base)
(vii) addition, substitution, elimination, condensation, hydrolysis
(viii) oxidation and reduction
[in equations for organic redox reactions, the symbols [O] and [H] are acceptable]
(c) interpret, and use the following terminology associated with organic reactivities:
(i) delocalisation
(ii) electronic effect (electron-donating and electron-withdrawing effect)
(iii) steric effect (steric hindrance)
(d) describe sp3 hybridisation, as in ethane molecule, sp2 hybridisation, as in ethene and benzene molecules, and sp hybridisation, as in ethyne molecule
(e) explain the shapes of, and bond angles in, the ethane, ethene, benzene, and ethyne molecules in relation to σ and π carbon-carbon bonds
(f) predict the shapes of, and bond angles in, molecules analogous to those specified in (e)
(g) apply (b) and (c) to the understanding of mechanisms in terms of organic structure and bonding
(h) recognise that the mechanisms of polar reactions involve the flow of electrons from electron-rich to electron-poor sites
11.2 Isomerism
Content
• Isomerism: constitutional (structural); cis-trans; enantiomerism
Learning Outcomes
Candidates should be able to:
(a) describe constitutional (structural) isomerism
(b) describe cis-trans isomerism in alkenes, and explain its origin in terms of restricted rotation due to the presence of π bonds
[use of E, Z nomenclature is not required]
(c) explain what is meant by a chiral centre
(d) deduce whether a given molecule is chiral based on the presence or absence of chiral centres and/or a plane of symmetry
(e) recognise that an optically active sample rotates plane-polarised light and contains chiral molecules
(f) recognise that enantiomers have identical physical properties except in the direction in which they rotate plane-polarised light
[usage of the term diastereomers is not required]
(g) recognise that enantiomers have identical chemical properties except in their interactions with another chiral molecule
(h) recognise that different stereoisomers exhibit different biological properties, for example in drug action
(i) deduce the possible isomers for an organic molecule of known molecular formula
(j) identify chiral centres and/or cis-trans isomerism in a molecule of given structural formula
In each of the sections below, 11.3 to 11.8, candidates will be expected to be able to predict the reaction products of a given compound in reactions that are chemically similar to those specified.
11.3 Hydrocarbons
Content
● Alkanes (exemplified by ethane)
(i) free-radical substitution reactions
● Alkenes (exemplified by ethene)
(i) electrophilic addition, including Markovnikov’s rule
(ii) reduction and oxidation reactions
● Arenes (exemplified by benzene and methylbenzene)
(i) influence of delocalised π electrons on structure and properties
(ii) electrophilic substitution reactions
(iii) oxidation of side-chain
● Hydrocarbons as fuels
Learning Outcomes
Candidates should be able to:
(a) explain the general unreactivity of alkanes, including towards polar reagents
(b) describe the chemistry of alkanes as exemplified by the following reactions of ethane:
(i) combustion
(ii) free-radical substitution by chlorine and by bromine
(c) describe the mechanism of free-radical substitution with particular reference to the initiation, propagation and termination reactions
(d) explain the general reactivity of alkenes towards electrophilic reagents/electrophiles
(e) describe the chemistry of alkenes as exemplified, where relevant, by the following reactions of ethene:
(i) electrophilic addition of water/steam, hydrogen halides and halogens
(ii) reduction via catalytic hydrogenation (catalytic addition of hydrogen; see also 8(j))
(iii) oxidation by cold, alkaline solution of manganate(VII) ions to form the diol
(iv) oxidation by hot, acidified solution of manganate(VII) ions leading to the rupture of the carbon-to-carbon double bond in order to determine the position of alkene linkages in larger molecules
(f) describe the mechanism of electrophilic addition in alkenes, using bromine with ethene as an example
(g) apply Markovnikov’s rule to the addition of hydrogen halides to unsymmetrical alkenes, and explain the composition of products in terms of the stability of carbocation intermediates
(h) explain, in terms of delocalisation of π electrons, the difference between benzene and alkene:
(i) reactivity towards electrophiles
(ii) preference of benzene to undergo substitution rather than addition reaction
(i) describe the chemistry of the benzene ring as exemplified by the following reactions of benzene and methylbenzene:
(i) electrophilic substitution reactions with chlorine and with bromine (recognise the use of Lewis acid as catalysts; see also Section 4)
(ii) nitration with concentrated nitric acid (recognise concentrated sulfuric acid as a Brønsted-Lowry acid catalyst; see also Section 4)
(iii) Friedel-Crafts alkylation with halogenoalkanes (recognise the use of Lewis acid as catalysts; see also Section 4)
(j) (i) describe the mechanism of electrophilic substitution in arenes, using the mono-bromination of benzene as an example
(ii) describe the effect of the delocalisation of electrons in arenes in such reactions
(k) describe the chemistry of the alkyl side-chain of benzene ring as exemplified by the following reactions of methylbenzene:
(i) free-radical substitution by chlorine and by bromine
(ii) complete oxidation to give benzoic acid
(l) predict whether halogenation will occur in the side-chain or aromatic nucleus in arenes depending on reaction conditions
(m) apply the knowledge of positions of substitution in the electrophilic substitution reactions of monosubstituted arenes
(n) recognise the environmental consequences of:
(i) carbon monoxide, oxides of nitrogen and unburnt hydrocarbons arising from the internal combustion engine and of their catalytic removal
(ii) gases that contribute to the enhanced greenhouse effect
(o) recognise that petroleum, a chemical feedstock, is a finite resource and the importance of recycling
11.4 Halogen Derivatives
Content
● Halogenoalkanes
(i) nucleophilic substitution
(ii) elimination
● Relative strength of the C-Hal bond
● Unreactivity of halogenoarenes
Learning Outcomes
Candidates should be able to:
(a) recall the chemistry of halogenoalkanes as exemplified by:
(i) the following nucleophilic substitution reactions of bromoethane: hydrolysis; formation of nitriles; formation of primary amines by reaction with ammonia
(ii) the elimination of hydrogen bromide from 2-bromopropane
(b) describe and explain the mechanisms of nucleophilic substitutions in halogenoalkanes:
(i) SN1, in terms of stability of the carbocation intermediates
(ii) SN2, in terms of steric hindrance in the halogenoalkanes
(c) explain the stereochemical outcome in nucleophilic substitution involving optically active substrates:
(i) inversion of configuration in SN2 mechanism
(ii) racemisation in SN1 mechanism
(d) interpret the different reactivities of halogenoalkanes, with particular reference to hydrolysis, and to the relative strengths of the C-Hal bonds
(e) explain the unreactivity of chlorobenzene compared to halogenoalkanes towards nucleophilic substitution, in terms of the delocalisation of the lone pair of electrons on the halogen and steric hindrance
(f) suggest characteristic reactions to differentiate between:
(i) different halogenoalkanes
(ii) halogenoalkanes and halogenoarenes
e.g. hydrolysis, followed by testing of the halide ions
(g) explain the uses of fluoroalkanes and fluorohalogenoalkanes in terms of their relative chemical inertness
(h) recognise the concern about the effect of chlorofluoroalkanes (CFCs) on the ozone layer
[the mechanistic details of how CFCs deplete the ozone layer are not required]
11.5 Hydroxy Compounds
Content
● Alcohols (exemplified by ethanol)
(i) formation of halogenoalkanes
(ii) reaction with sodium; oxidation; dehydration
(iii) the tri-iodomethane test
● Phenol
(i) its acidity; reaction with bases and sodium
(ii) nitration of, and bromination of, the aromatic ring
Learning Outcomes
Candidates should be able to:
(a) recall the chemistry of alcohols, exemplified by ethanol:
(i) combustion
(ii) nucleophilic substitution to give halogenoalkanes
(iii) reaction with sodium
(iv) oxidation to carbonyl compounds and carboxylic acids
(v) dehydration to alkenes
(b) suggest characteristic distinguishing reactions for the different classes of alcohols (primary, secondary and tertiary alcohols), e.g. mild oxidation
(c) deduce the presence of a CH3CH(OH)– group in an alcohol from its reaction with alkaline aqueous iodine to form tri-iodomethane
(d) recall the chemistry of phenol, as exemplified by the following reactions:
(i) with bases
(ii) with sodium
(iii) nitration of, and bromination of, the benzene ring
(e) explain the relative acidities of water, phenol and ethanol in aqueous medium (interpret as Brønsted-Lowry acids)
11.6 Carbonyl Compounds
Content
● Aldehydes (exemplified by ethanal)
(i) oxidation to carboxylic acid
(ii) nucleophilic addition with hydrogen cyanide
(iii) characteristic tests for aldehydes
● Ketones (exemplified by propanone and phenylethanone)
(i) nucleophilic addition with hydrogen cyanide
(ii) characteristic tests for ketones
Learning Outcomes
Candidates should be able to:
(a) describe the formation of aldehydes and ketones from, and their reduction to, primary and secondary alcohols respectively
(b) describe the mechanism of the nucleophilic addition reactions of hydrogen cyanide with aldehydes and ketones
(c) explain the differences in reactivity between carbonyl compounds and alkenes towards nucleophilic reagents, such as lithium aluminium hydride and hydrogen cyanide
(d) describe the use of 2,4-dinitrophenylhydrazine (2,4-DNPH) to detect the presence of carbonyl compounds
(e) deduce the nature (aldehyde or ketone) of an unknown carbonyl compound from the results of simple tests (i.e. Fehling’s and Tollens’ reagents; ease of oxidation)
(f) deduce the presence of a CH3CO– group in a carbonyl compound from its reaction with alkaline aqueous iodine to form tri-iodomethane
11.7 Carboxylic Acids and Derivatives
Content
● Carboxylic acids (exemplified by ethanoic acid and benzoic acid)
(i) formation from primary alcohols and nitriles
(ii) salt, ester and acyl chloride formation
● Acyl chlorides (exemplified by ethanoyl chloride)
(i) ease of hydrolysis compared with alkyl and aryl chlorides
(ii) reaction with alcohols, phenols and primary amines
● Esters (exemplified by ethyl ethanoate and phenyl benzoate)
(i) formation from carboxylic acids and from acyl chlorides
(ii) hydrolysis (under acidic and under basic conditions)
Learning Outcomes
Candidates should be able to:
(a) describe the formation of carboxylic acids from alcohols, aldehydes and nitriles
(b) describe the reactions of carboxylic acids in the formation of:
(i) salts
(ii) esters on condensation with alcohols, using ethyl ethanoate as an example
(iii) acyl chlorides, using ethanoyl chloride as an example
(iv) primary alcohols, via reduction with lithium aluminium hydride, using ethanol as an example
(c) explain the acidity of carboxylic acids and of chlorine-substituted ethanoic acids in terms of their structures
(d) describe the hydrolysis of acyl chlorides
(e) describe the condensation reactions of acyl chlorides with alcohols, phenols and primary amines
(f) explain the relative ease of hydrolysis of acyl chlorides, alkyl chlorides and aryl chlorides
(g) describe the formation of esters from the condensation reaction of acyl chlorides, using phenyl benzoate as an example
(h) describe the acid and base hydrolysis of esters
11.8 Nitrogen Compounds
Content
● Amines (exemplified by ethylamine and phenylamine)
(i) their formation
(ii) salt formation
(iii) other reactions of phenylamine
● Amides (exemplified by ethanamide)
(i) formation from acyl chlorides
(ii) neutrality of amides
(iii) hydrolysis (under acidic and under basic conditions)
● Amino acids (exemplified by aminoethanoic acid)
(i) their acid and base properties
(ii) zwitterion formation
● Proteins
(i) formation of proteins
(ii) hydrolysis of proteins
Learning Outcomes
Candidates should be able to:
(a) describe the formation of amines as exemplified by ethylamine (through amide and nitrile reduction; see also Section 11.4) and by phenylamine (through the reduction of nitrobenzene)
(b) describe the reaction of amines in the formation of salts
(c) describe and explain the basicity of primary, secondary and tertiary amines in the gaseous phase (interpret as Lewis bases)
(d) explain the relative basicities of ammonia, ethylamine and phenylamine in aqueous medium, in terms of their structures
(e) describe the reaction of phenylamine with aqueous bromine
(f) describe the formation of amides from the condensation reaction between RNH2 and R'COCl
(g) explain why an amide is neutral in terms of delocalisation of the lone pair of electrons on nitrogen
(h) describe the chemistry of amides, exemplified by the following reactions:
(i) hydrolysis on treatment with aqueous alkali or acid
(ii) reduction to amines with lithium aluminium hydride
(i) describe the acid/base properties of amino acids and the formation of zwitterions [knowledge of isoelectric points is not required]
(j) describe the formation of peptide (amide) bonds between α-amino acids, and hence explain protein formation
(k) describe the hydrolysis of proteins
12. Electrochemistry
Content
● Redox processes: electron transfer and changes in oxidation number (oxidation state)
● Electrode potentials
(i) standard electrode (redox) potentials, E⦵; the redox series
(ii) standard cell potentials, E⦵ cell, and their uses
(iii) batteries and fuel cells
● Electrolysis
(i) factors affecting the amount of substance liberated during electrolysis
(ii) the Faraday constant; the Avogadro constant; their relationship
(iii) industrial uses of electrolysis
Learning Outcomes
Candidates should be able to:
(a) describe and explain redox processes in terms of electron transfer and/or of changes in oxidation number (oxidation state)
(b) define the terms:
(i) standard electrode (redox) potential
(ii) standard cell potential
(c) describe the standard hydrogen electrode
(d) describe methods used to measure the standard electrode potentials of:
(i) metals or non-metals in contact with their ions in aqueous solution
(ii) ions of the same element in different oxidation states
(e) calculate a standard cell potential by combining two standard electrode potential
(f) use standard cell potentials to:
(i) explain/deduce the direction of electron flow from a simple cell
(ii) predict the spontaneity of a reaction
(g) understand the limitations in the use of standard cell potentials to predict the spontaneity of a reaction
(h) construct redox equations using the relevant half-equations (see also Section 13)
(i) state and apply the relationship ΔG⦵ = –nFE⦵ to electrochemical cells, including the calculation of E⦵ for combined half reactions
(j) predict qualitatively how the value of an electrode potential varies with the concentration of the aqueous ion
(k) state the possible advantages of developing other types of cell, e.g. the H2/O2 fuel cell and improved batteries (as in electric vehicles) in terms of smaller size, lower mass and higher voltage
(l) state the relationship, F = Le, between the Faraday constant, the Avogadro constant and the charge on the electron
(m) predict the identity of the substance liberated during electrolysis from the state of electrolyte (molten or aqueous), position in the redox series (electrode potential) and concentration
(n) calculate:
(i) the quantity of charge passed during electrolysis
(ii) the mass and/or volume of substance liberated during electrolysis
(o) explain, in terms of the electrode reactions, the industrial processes of:
(i) the anodising of aluminium
(ii) the electrolytic purification of copper [technical details are not required]
13. An Introduction to the Chemistry of Transition Elements
Content
• General physical and characteristic chemical properties of the first set of transition elements, titanium to copper
• Colour of complexes
Learning Outcomes
Candidates should be able to:
(a) explain what is meant by a transition element, in terms of d block elements forming one or more stable ions with partially filled d subshells
(b) state the electronic configuration of a first row transition element and its ions
(c) explain why atomic radii and first ionisation energies of the transition elements are relatively invariant
(d) contrast, qualitatively, the melting point and density of the transition elements with those of calcium as a typical s block element
(e) describe the tendency of transition elements to have variable oxidation states
(f) predict from a given electronic configuration, the likely oxidation states of a transition element
(g) describe and explain the use of Fe3+/Fe2+, MnO4–/Mn2+ and Cr2O72–/Cr3+ as examples of redox systems (see also Section 12)
(h) predict, using E⦵ values, the likelihood of redox reactions
(i) define the terms ligand and complex as exemplified by the complexes of copper(II) ions with water, ammonia and chloride ions as ligands (including the transition metal complexes found in the Qualitative Analysis Notes)
(j) explain qualitatively that ligand exchange may occur, as exemplified by the formation of the complexes in (i), including the colour changes involved, and CO/O2 exchange in haemoglobin
(k) describe, using the shape and orientation of the d orbitals, the splitting of degenerate d orbitals into two energy levels in octahedral complexes
(l) explain, in terms of d orbital splitting and d-d transition, why transition element complexes are usually coloured
[knowledge of the relative order of ligand field strength is not required]
(m) explain how some transition elements and/or their compounds can act as catalysts (see also 8(j))
Practical Assessment
Scientific subjects are, by their nature, experimental. It is therefore important that, wherever possible, the candidates carry out appropriate practical work to support the learning of this subject and to develop the expected practical skills.
Paper 4 Practical
This paper is designed to assess a candidate's competence in those practical skills which can realistically be assessed within the context of a formal practical assessment.
Candidates will be assessed in the following skill areas:
(a) Planning (P)
Candidates should be able to:
● define the question/problem using appropriate knowledge and understanding
● give a clear logical account of the experimental procedure to be followed
● describe how the data should be used in order to reach a conclusion
● assess the risks of the experiment and describe precautions that should be taken to keep risks to a minimum
(b) Manipulation, measurement and observation (MMO)
Candidates should be able to:
● demonstrate a high level of manipulative skills in all aspects of practical activity
● make and record accurate observations with good details and measurements to an appropriate degree of precision
● make appropriate decisions about measurements or observations
● recognise anomalous observations and/or measurements (where appropriate) with reasons indicated
(c) Presentation of data and observations (PDO)
Candidates should be able to:
● present all information in an appropriate form
● manipulate measurements effectively in order to identify trends/patterns
● present all quantitative data to an appropriate number of decimal places/significant figures
(d) Analysis, conclusions and evaluation (ACE)
Candidates should be able to:
● analyse and interpret data or observations appropriately in relation to the task
● draw conclusion(s) from the interpretation of experimental data or observations and underlying principles
● make predictions based on their data and conclusions
● identify significant sources of errors, limitations of measurements and/or experimental procedures used, and explain how they affect the final result(s)
● state and explain how significant errors/limitations may be overcome or reduced, as appropriate, including how experimental procedures may be improved
One, or more, of the questions may incorporate some assessment of Skill P, set in the context of the syllabus content, requiring candidates to apply and integrate knowledge and understanding from different sections of the syllabus. It may also require the treatment of given experimental data to draw a relevant conclusion and analyse a proposed plan.
The assessment of skills MMO, PDO and ACE will also be set mainly in the context of the syllabus content.
The assessment of PDO and ACE may also include questions on data-analysis which do not require
practical equipment and apparatus.
Within the Scheme of Assessment, Paper 4 is weighted to 20% of the Higher 2 assessment. It is therefore recommended that the schemes of work include learning opportunities that apportion a commensurate amount of time for the development and acquisition of practical skills. The guidance for practical work, which is published separately, will provide examples of appropriate practical activities.
Candidates should be able to use appropriate apparatus/equipment to record a range of measurements such as mass, time, volume and temperature. In addition, candidates are expected to have been exposed to a range of experimental techniques in the following areas:
1) Titration, e.g. acid-base titration (with suitable indicators such as methyl orange, screened methyl orange, thymolphthalein and thymol blue), redox titration, iodimetric titration, indirect titration, including the preparation of standard solutions. Other types of titrations may also be required, where appropriate, sufficient working details will be given.
2) Gravimetric analysis, e.g. volatilisation gravimetry
3) Gas collection
4) Thermochemistry, including thermometric titration
5) Chemical kinetics, e.g. continuous and initial rate methods
6) Qualitative inorganic analysis involving an element, a compound or a mixture. Systematic analysis will not be required. Candidates should be familiar with the reactions of cations, reactions of anions and test for gases as detailed in the Qualitative Analysis Notes.
Candidates would not be required to carry out tests involving hexane, sulfur dioxide gas, nitrite ions or sulfite ions.
Reactions involving ions not included in the Qualitative Analysis Notes may be tested: in such cases, candidates will not be expected to identify the ions but only to draw conclusions of a general nature.
Candidates should not attempt tests, other than those specified, on substances, except when it is appropriate to test for a gas.
7) Qualitative organic analysis requiring a knowledge of simple organic reactions as outlined in Section 11.3 to 11.8, e.g. test-tube reactions indicating the presence of unsaturation (C=C), alcoholic, phenolic, carbonyl, carboxyl and amino groups, may be set, but this would be for the testing of observation skills and drawing general conclusions only.
8) Simple organic synthesis and purification, including use of water bath, setting up and use of reflux and distillation apparatus. Conducting a large-scale organic synthesis involving reflux, distillation and purification would not be expected during the practical examination.
This is not intended to be an exhaustive list.
Candidates are not allowed to refer to notebooks, textbooks or any other information in the practical examination. Qualitative Analysis Notes will be included in the question paper for the use of candidates in the examination.
Apparatus List
This list given below has been drawn up in order to give guidance to Centres concerning the apparatus that is expected to be generally available for examination purposes. The list is not intended to be exhaustive; in particular, items (such as Bunsen burners, tripods) that are commonly regarded as standard equipment in a chemical laboratory are not included.
Unless otherwise stated, the rate of allocation is “per candidate”.
two burettes, 50 cm3
two pipettes, 25 cm3
one pipette, 10 cm3
teat/dropping pipettes
one pipette filler
two conical flasks within the range 150 cm3 to 250 cm3
third conical flask within the range 150 cm3 to 250 cm3, with suitable bung and delivery tube
tub suitable for acting as a trough (for collecting gas over water)
volumetric flask, 250 cm3
two measuring cylinders, 10 cm3
measuring cylinders, 25 cm3 and 50 cm3
two filter funnels
two porcelain crucibles, approximately 15 cm3, with lids
one pipe-clay triangle
one evaporating basin, at least 30 cm3
beakers, squat form with lip: 100 cm3, 250 cm3
thermometers: –10 °C to +110 °C at 1 °C
–5 °C to +50 °C at 0.2 °C
a polystyrene, or other plastic beaker of approximate capacity 150 cm3
test-tubes (Pyrex or hard glass), approximately 125 mm × 15 mm
boiling tubes, approximately 150 mm × 25 mm
stopwatch to measure to an accuracy of about 1 s
balance, single-pan, direct reading, 0.01 g or better (1 per 8–12 candidates)
stand and clamp suitable for a burette
wash bottle
The apparatus and material requirements for Paper 4 will vary year on year. Centres will be notified in advance of the details of the apparatus and materials required for each practical examination.
Reagents
This list given below has been drawn up in order to give guidance to Centres concerning the standard reagents that are expected to be generally available for examination purposes. The list is not intended to be exhaustive and Centres will be notified in advance of the full list of all the reagents that are required for each practical examination.
aqueous sodium hydroxide (approximately 2.0 mol dm–3)
aqueous ammonia (approximately 2.0 mol dm–3)
hydrochloric acid (approximately 2.0 mol dm–3)
nitric acid (approximately 2.0 mol dm–3)
sulfuric acid (approximately 1.0 mol dm–3)
aqueous silver nitrate (approximately 0.05 mol dm–3)
aqueous barium nitrate (approximately 0.2 mol dm–3)
limewater (a saturated solution of calcium hydroxide)
aqueous potassium manganate(VII) (approximately 0.02 mol dm–3)
aqueous potassium iodide (approximately 0.1 mol dm–3)
aluminium foil
red and blue litmus paper or Universal Indicator paper

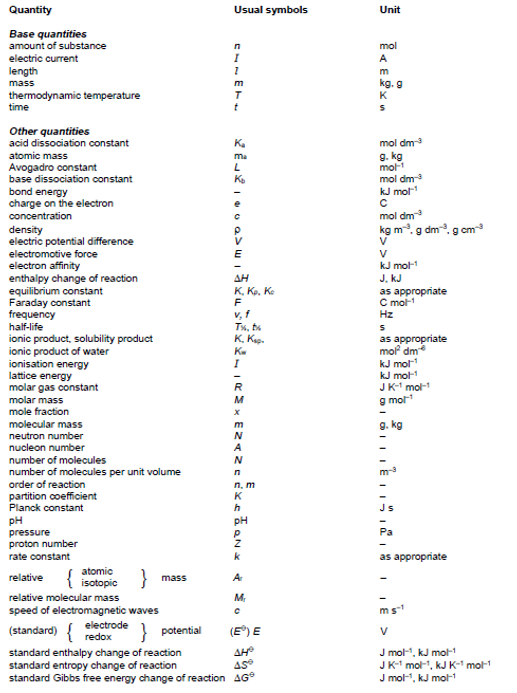
Summary of Key Quantities and Units
The list below is intended as a guide to the more important quantities which might be encountered in
teaching and used in question papers. The list is not exhaustive.
Mathematical Requirements
It is assumed that candidates will be competent in the techniques described below.
Make calculations involving addition, subtraction, multiplication and division of quantities.
Make approximate evaluations of numerical expressions.
Express small fractions as percentages, and vice versa.
Calculate an arithmetic mean.
Transform decimal notation to power of ten notation (standard form).
Use calculators to evaluate logarithms, squares, square roots, and reciprocals.
Change the subject of an equation. (Most such equations involve only the simpler operations but may include positive and negative indices and square roots.)
Substitute physical quantities into an equation using consistent units so as to calculate one quantity. Check the dimensional consistency of such calculations, e.g. the units of a rate constant k.
Solve simple algebraic equations.
Comprehend and use the symbols/notations <, >, ≈, /, Δ, ≡, x (or <x>).
Test tabulated pairs of values for direct proportionality by a graphical method or by constancy of ratio.
Select appropriate variables and scales for plotting a graph, especially to obtain a linear graph of the form y = mx + c.
Determine and interpret the slope and intercept of a linear graph.
Choose by inspection a straight line that will serve as the ‘least bad’ linear model for a set of data presented graphically.
Understand (i) the slope of a tangent to a curve as a measure of rate of change, (ii) the ‘area’ below a curve where the area has physical significance, e.g. Boltzmann distribution curves.
Comprehend how to handle numerical work so that significant figures are neither lost unnecessarily nor used beyond what is justified.
Estimate orders of magnitude.
Formulate simple algebraic equations as mathematical models, e.g. construct a rate equation, and identify failures of such models.
Calculators
Any calculator used must be on the Singapore Examinations and Assessment Board list of approved calculators.
Glossary of Terms
It is hoped that the glossary (which is relevant only to science subjects) will prove helpful to candidates as a guide, i.e. it is neither exhaustive nor definitive. The glossary has been deliberately kept brief not only with respect to the number of terms included but also to the descriptions of their meanings. Candidates should appreciate that the meaning of a term must depend in part on its context.
1. Define (the term(s)...) is intended literally, only a formal statement or equivalent paraphrase being required.
2. What do you understand by/What is meant by (the term(s)...) normally implies that a definition should be given, together with some relevant comment on the significance or context of the term(s) concerned, especially where two or more terms are included in the question. The amount of supplementary comment intended should be interpreted in the light of the indicated mark value.
3. State implies a concise answer with little or no supporting argument, e.g. a numerical answer that can be obtained ‘by inspection’.
4. List requires a number of points, generally each of one word, with no elaboration. Where a given number of points is specified, this should not be exceeded.
5. Explain may imply reasoning or some reference to theory, depending on the context.
6. Describe requires candidates to state in words (using diagrams where appropriate) the main points of the topic. It is often used with reference either to particular phenomena or to particular experiments. In the former instance, the term usually implies that the answer should include reference to (visual) observations associated with the phenomena.
In other contexts, describe and give an account of should be interpreted more generally, i.e. the candidate has greater discretion about the nature and the organisation of the material to be included in the answer. Describe and explain may be coupled in a similar way to state and explain.
7. Discuss requires candidates to give a critical account of the points involved in the topic.
8. Outline implies brevity, i.e. restricting the answer to giving essentials.
9. Predict implies that the candidate is not expected to produce the required answer by recall but by making a logical connection between other pieces of information. Such information may be wholly given in the question or may depend on answers extracted in an early part of the question.
10. Deduce is used in a similar way as predict except that some supporting statement is required, e.g. reference to a law/principle, or the necessary reasoning is to be included in the answer.
11. Comment is intended as an open-ended instruction, inviting candidates to recall or infer points of interest relevant to the context of the question, taking account of the number of marks available.
12. Suggest is used in two main contexts, i.e. either to imply that there is no unique answer (e.g. in chemistry, two or more substances may satisfy the given conditions describing an ‘unknown’), or to imply that candidates are expected to apply their general knowledge to a ‘novel’ situation, one that may be formally ‘not in the syllabus’.
13. Find is a general term that may variously be interpreted as calculate, measure, determine, etc.
14. Calculate is used when a numerical answer is required. In general, working should be shown, especially where two or more steps are involved.
15. Measure implies that the quantity concerned can be directly obtained from a suitable measuring instrument, e.g. length, using a rule, or angle, using a protractor.
16. Determine often implies that the quantity concerned cannot be measured directly but is obtained by calculation, substituting measured or known values of other quantities into a standard formula, e.g. relative molecular mass.
17. Estimate implies a reasoned order of magnitude statement or calculation of the quantity concerned, making such simplifying assumptions as may be necessary about points of principle and about the values of quantities not otherwise included in the question.
18. Sketch, when applied to graph work, implies that the shape and/or position of the curve need only be qualitatively correct, but candidates should be aware that, depending on the context, some quantitative aspects may be looked for, e.g. passing through the origin, having an intercept, asymptote or discontinuity at a particular value.
In diagrams, sketch implies that a simple, freehand drawing is acceptable: nevertheless, care should be taken over proportions and the clear exposition of important details.
19. Construct is often used in relation to chemical equations where a candidate is expected to write a balanced equation, not by factual recall but by analogy or by using information in the question.
20. Compare requires candidates to provide both the similarities and differences between things or concepts.
21. Classify requires candidates to group things based on common characteristics.
22. Recognise is often used to identify facts, characteristics or concepts that are critical (relevant/appropriate) to the understanding of a situation, event, process or phenomenon.
Textbooks and References
Teachers may find reference to the following books helpful.
Cambridge International AS and A Level Chemistry by Peter Cann and Peter Hughes, published by Hodder Education
Cambridge International AS and A Level Chemistry Coursebook with CD-ROM (2nd Edition) by Lawrie Ryan and Roger Norris, published by Cambridge University Press
A-Level Chemistry (4th Edition) by E N Ramsden, published by Oxford University Press
Understanding Chemistry for Advanced Level (3rd Edition), by Ted Lister and Janet Renshaw, published by Oxford University Press
AS and A Level Chemistry through Diagrams by Michael Lewis, published by Oxford University Press
Chemistry in Context (6th Edition) by Graham Hill and John Holman, published by Oxford University Press
Chemistry in Context Laboratory Manual and Study Guide (5th Edition) by Graham Hill and John Holman,
published by Oxford University Press
Experiments and Exercises in Basic Chemistry (7th Edition) by Steve Murov and Brian Stedjee, published by Wiley
Chemical Ideas (Salters Advanced Chemistry) by Adelene Cogill, et al., published by Pearson Education Limited
The Language of Mathematics in Science: A Guide for Teachers of 11–16 Science by R Boohan, published by the Association for Science Education
Teachers are encouraged to choose texts for class use which they feel will be of interest to their students and will support their own teaching style.
Many publishers are also producing videos and software appropriate for A-Level Chemistry students.
Data Booklet
Contents: Tables of Chemical data
1 Important values, constants and standards
2 Ionisation energies (1st, 2nd, 3rd and 4th) of selected elements in kJ mol–1
3 Bond energies
4 Standard electrode potential and redox potentials, E⦵ at 298 K (25 °C)
5 Atomic and ionic radii
6 Typical proton (1H) chemical shift values (δ) relative to TMS = 0
7 Characteristic infra-red absorption frequencies for some selected bonds
8 The orientating effect of groups in aromatic substitution reactions
9 Qualitative Analysis Notes
10 The Periodic Table of Elements

















bottom of page